Thermo Genesis Holdings THMO
Thanks to a Reader for sending this..A Small Biotec Company
This looks very interesting and could be a Game Changer !
ThermoGenesis Holdings, Inc. (THMO), a market leader in automated cell processing tools and services in the cell and gene therapy field, announced today that on Wednesday April 15th, following submission of notification to the U.S. Food and Drug Administration (FDA), the Company has received the necessary acknowledgement letter which provided confirmation that the ThermoGenesis’ SARS-CoV-2 (COVID-19) IgM/IgG Antibody Fast Detection Kit has been appropriately validated in accordance with Section IV.D. of the “Policy for Diagnostic Tests for Coronavirus Disease – 2019 during the Public Health Emergency,” issued by FDA on March 16, 2020. The required registrations and listings with FDA have been completed, which now allow the diagnostic kit to be distributed.
An informational presentation on the COVID-19 Kit is available on the Company’s website. The presentation is for educational purposes and will allow users to better understand how the antibody test works and how to evaluate the results. Please visit the link below:
· https://thermogenesis.com/rapid-covid-19-point-of-care-diagnostic-test/
As previously announced, the COVID-19 Kit will be jointly marketed by ThermoGenesis and ImmuneCyte Life Sciences, Inc. Inquiries from health professionals and the media about the Kit should be directed via email to ir@thermogenesis.com.
WOW…AMERICAN INGENUITY ALIVE AND WELL
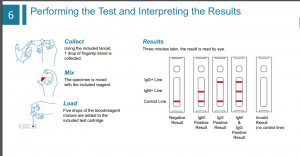
• Human immune system mounts
an IgM and IgG antibody response
in patients infected with
coronavirus
• IgM and IgG antibodies can be
detected in Covid-19 infected
patients 0-7 days earlier than
clinical signs and symptoms
appear.
• ThermoGenesis Covid-19 IgM/IgG
Detection Kit is administered
similar to a home diabetic glucose
test & can be read within 10
minutes.
• If detected in asymptomatic
person, it may also mean the
person has recovered from mild
infection and have developed
protective immunity.
111 of 112 PCR-NEGATIVE PATIENTS TESTED NEGATIVE (99.1% Negative Coincidence Rate)
14 of 14 PCR-POSITIVE PATIENTS TESTED POSITIVE (100% Positive Coincidence Rate)
In additional studies, no cross-reactivity (false positives) was observed in PCR-negative patients
known to be positive for Influenza A, Influenza B, Adenovirus, Mycoplasma pneumonia, or
Respiratory syncytial virus infections.